| Desirable Properties of Sizing Materials
|
| The sizing material must fulfil some essential properties and at the same time it is expected that it will have some additional desirable properties. The sizing material must form a smooth and uniform coating the yarn surface. This is known as film forming property. The coating will not only embed the protruding fibres (hairs) on the yarn body but also protect the yarn structure from repeated abrasion during weaving. The size film should adhere with the fibres strongly to prevent shedding (dropping of size film). The film should also have enough flexibility to cope with the flexing or bending of yarns around the back rest, heald eyes and other loom components. The non-exhaustive list of essential and desirable properties is given in Table 4.1.
|
Table 4.1: Essential and desirable properties of sizing materials
|
| Film forming |
Controllable viscosity |
| Adhesion |
Easy removal and recyclability |
| Optimum penetration |
Neutral pH |
| Film flexibility and elasticity |
Non-polluting |
| Lubrication |
Cheap |
| Bacterial resistance |
|
|
The specific requirements of sizing material properties will depend on the following factors.
|
- Type of fibre (cotton, viscose, nylon, polyester etc.)
- Type of yarn (ring, rotor, friction, air-jet etc.)
- Type of loom (shuttle, projectile, air-jet, water jet etc)
- Construction features (weave, yarn counts and sett)
|
Composition of Sizing Material
|
The specific composition of sizing material depends on the fibre type, yarn type, yarn count, fabric sett etc. However, the materials can be classified under the categories of adhesive, softening agent, antimicrobial agent etc. The adhesive part is responsible for forming the film and adhering with the fibres. Softening agent makes the film flexible so that the film can bend easily without forming cracks. Antimicrobial agents are added to thwart the mildew to grow on the size film. Sometimes, weighting agents and dyes are also added to fulfil specific requirements.
|
Cotton yarns, in general, are sized by the starch which forms the adhesive component of the size mix. The reason behind the popularity of starch can be attributed to the following factors. |
- Starch is chemically same with cotton and rayon and thus the adhesion is very good.
- Desizing is easy
- Relatively cheap
- Properties can be tuned to cope with the need
|
However, starch gives very stiff film. It has higher biological oxygen demand (BOD). Besides, cooking of starch is required to attain uniformity. Besides, starch has poor bacterial resistance. Overall, the positive attributes of starch dominates over the drawbacks and thus it is still being used in the industry as the primary materials for the sizing of cotton yarns.
|
| The softening materials compensates for the abrasive and harsh feel that is provided by most of the starches. Softeners also lubricate the yarns so that they can pass easily over machines parts without shedding. It also prevents the sticking of size ingredients over the drying cylinders. Mutton tallow which is composed of glycerides of palmitic, stearic and oleic acids is used as softeners. The proportion of softener in the size mix is very crucial as excess use of it deteriorates the strength of size film.
|
| Sources of Starch
|
| Starches are available from the seed, root or pith of plants. Corn, rice and wheat are the examples of seed starch. Potato and Tapioca starches are obtained from roots. Sago starch is obtained from pith. Starches are prepared by grinding the seed, root or pith into fine flour. When the flour is mixed with water and cooked, it produces a thick and smooth glutinous solution. Corn (Maize) starch is the most popular type of starch used in textile sizing. Around 50% of the corn is composed of starch. Corn starch is generally preferred for the sizing of coarse and medium count yarns. Potato yields around 20% starch. It is slow congealing type and therefore gets more chance to penetrate within the yarn structure. It forms a smooth and pliable film on the yarn body. Potato starch is preferred for sizing finer yarns.
|
Chemical Structure of Starch
|
| Cellulose and Starch |
- Chemically same
- Both are polymer of glucose
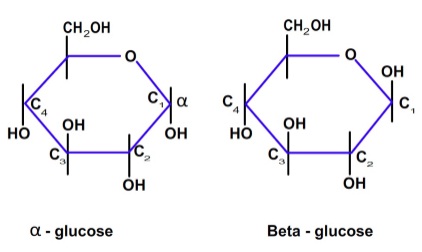
- Glucose can have two structural (anomeric) form known as α and β (Figure 4.3).
|
| 
|
Figure 4.3: Structure of α and β glucose
|
| It can be seen from Figure 4.3 that the position of OH group in the pyranose ring at C2, C3 and C4 are same in cellulose and starch. For α -D-glucose, which is the building block of starch, both the OH groups are down with respect to the next glucose molecule. Therefore no change in orientation is needed for condensation as shown in Figure 4.4.
|
|
Figure 4.4: Condensation of α-glucose units |
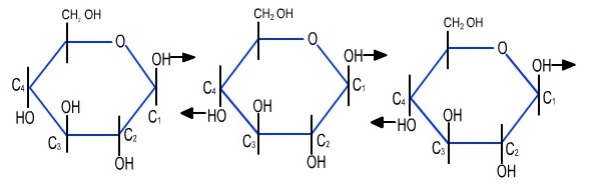
On the other hand, in β glucose, the OH group is up at C1 and down at C4. As a result, every other glucose unit must flip over before the 1,4 hydroxyls can come closer for condensation. This is shown in Figure 4.5.
|
|
Figure 4.5: Condensation of β-glucose units
|
| |
Starches are having two components. The straight chain component is called amylose. The branched chain component is called amylopectin. The differences between amylose and amylopectin have been highlighted in Table 4.2. |
Table 4.2: Differences between amylose and amylopectin |
| Amylose |
Amylopectin |
| Provides strength |
Prevents rapid gelling |
| Water soluble |
Water insoluble |
| Low molecular weight |
Relatively high molecular weight |
| 20-30% |
70-80% |
|
The proportion of amylose and amylopection differs depending on source. For example, in potato starch the ratio is 20:80 whereas in wheat starch the ratio is 25: 75. |
|
Starch remains tightly bound in granules and therefore it does not act as adhesive in cold water. Cooking of starch is required to make it soluble in water. The change in viscosity of starch solution during cooking is shown in Figure 4.6.
|
|
Figure 4.6: Cooking of starch
|
Within the granule, the chain molecules of amylose and amylopection are arranged radially in stratified layers. External heat energy is required for the penetration of water molecule within the structure. The temperature at which the thermal energy becomes sufficient to overcome hydrogen bonding within the structure is called ‘gelatinisation’ temperature (P). Crystallization of starch is lost during gelation. As the water penetrates, the chain molecules are pushed away from each other causing swelling of the starch granule. This is marked by increased in viscosity of the solution (Q). This continues up to the point R. Aided by the continuous shearing provided by the stirring, the starch granules finally break. The chain molecules of amylose and amylopection come out within the solution causing reduction in viscosity (T). When all the granules have burst, the viscosity stabilizes or levels off (T). When the solution is cooled, the starch gels due to the formation of a rigid interlocked micelle-like structure having hydrogen bonding (U). This gel form of starch can form a continuous coating on the yarn surface.
|
|
The viscosity of the sizing paste influences the wet pick-up and resultant add-on %. The viscosity is influenced by the concentration of starch (solid content) and molecular chain length of starch. To reduce the concentration of the sizing paste, keeping the solid content same, acid treatment is performed. Aqueous solution of starch is treated with hydrochloric acids at specified temperature and duration. The acid cleaves the polymer at the glycoside linkage and thus the length of the polymer chain is reduced (Figure 4.7). Hence, the viscosity is reduced and the fluidity, which is the reciprocal of viscosity, increases. The acid treated starch is often termed as ‘thin boiling starch’ as it results lower viscosity than the normal starch at a given concentration.
|
|
Figure 4.7: Acid treatment of starch to reduce the molecular chain length
|
|